Research at the Johns Hopkins School of Nursing is coordinated between the Nursing Office of Research Administration (NORA), Office of Finance and Administration, and Johns Hopkins University Research Administration as well as Johns Hopkins Medical Institutes.
The NORA collaborates with:
JHSON department chairs and Center faculty
Office of Finance and Administration (SrRSAs)
Office of Development and Alumni Relations (DAR)
Johns Hopkins University Research Administration (JHURA)
Johns Hopkins Institute for Clinical and Translational Research (ICTR)
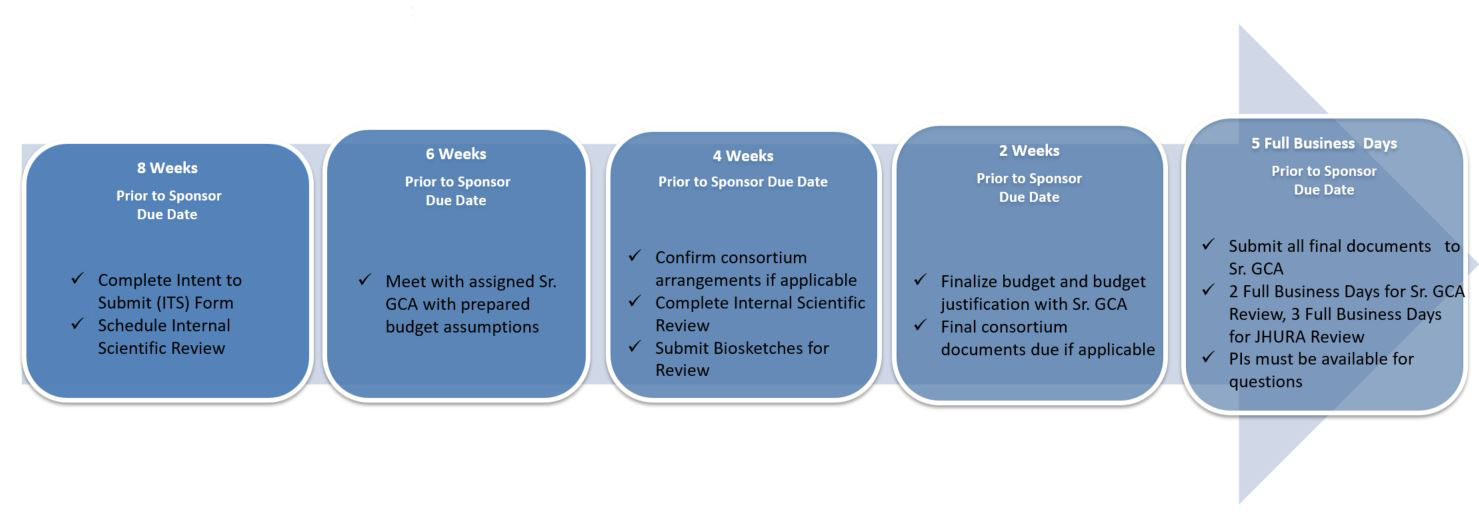
Pre-Award Proposal Processing:
*Internal JHU submissions require an Intent to Submit but do not need to be routed through JHURA for approval or submission; the PI submits the proposal. PI’s are encouraged to work with their SrRSA to develop any budget materials and they will assist in setting up the account and all monitoring and post-award activities should an award be funded.
The School of Nursing Strongly encourages the Internal Review Process.
Internal Scientific Reviews enhance and improve proposals and increase the likelihood of successful award applications by leveraging our internal expertise to evaluate the key components and how they are presented to address the outlined goals. All JH School of Nursing applications to external sponsors (extramural), excluding incoming sub-awards/subcontracts, are required to receive an internal scientific review.
Additional Resources
Johns Hopkins University Research Administration
JHURA supports the following functions:
Limited Submission Funding Opportunities
Proposal Facilitation and Submission
Award Negotiations and Acceptance
Subawards Execution and Monitoring
Collaborations (MOUs, Educational Agreements, etc.)
Communication and Outreach
Coeus System Administration
Data Monitoring and Reporting
Training and Education
Compliance
Nursing Office of Research Administration (NORA) was developed to facilitate excellence in nursing research. The goal of the NORA is to facilitate faculty and student research by offering programs and resources to support the development, submission, conduct and publication of research; and by providing the needed information technology to foster research. The NORA works in collaboration with the School of Nursing department chairs and Center faculty to provide a supportive environment for faculty, students, and staff involved in research.
HUDMS fosters data management best practices by providing guidance, education and training and offering data archiving and sharing services in support of researcher, institution, and agency goals.
The Johns Hopkins Medicine Institutional Review Boards (JHM IRBs) are responsible for protecting the rights and welfare of the human subjects of research conducted by faculty and staff at the Institutions. The JHM IRBs review all human subjects research projects conducted by Hopkins faculty and staff.
Johns Hopkins Technology Ventures helps develop new discoveries and inventions into products and services that benefit society and transform the world. We help inventors and entrepreneurs protect and license their innovations and foster collaboration with business partners who specialize in launching new inventions.
Our mission is to protect the intellectual property of Johns Hopkins faculty and to commercialize these inventions for the public good and bring the benefits of discovery to the world.
The Johns Hopkins Institute for Clinical and Translational Research (ICTR), established in 2007, is one of more than 60 medical research institutions working together as a national consortium to improve the way biomedical research is conducted across the country.
The ICTR addresses obstacles in translating basic science discoveries into research in humans, translating clinical discoveries into the community and communicating experience from clinical practice back to researchers.
The Women’s Health Research Group began as an interdisciplinary group of faculty from the Johns Hopkins Schools of Medicine, Public Health and Nursing who sought to promote collaborative research on women’s health and gender differences in health across the three institutions. Launched in 2008, they host networking
sessions throughout the academic year that are open to faculty and trainees at all levels, and an annual symposium that highlights new research from junior faculty and trainees.To be added to the WHRG email list, click here.